|
In almost every video and webpage I visit, I see people claiming that J. Because like charges repel each other and opposite charges attract, Thomson concluded that the particles had a net negative charge these particles are now called electrons.I am trying to make a quick sanity check. These particles were emitted by the negatively charged cathode and repelled by the negative terminal of an electric field. More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. He demonstrated that cathode rays could be deflected, or bent, by magnetic or electric fields, which indicated that cathode rays consist of charged particles (Figure 1.17c). Thomson (1856–1940) proved that atoms were not the most basic form of matter. 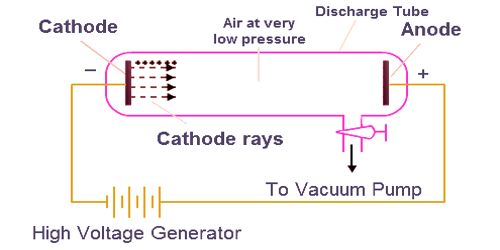
The cathode ray tube was first invented by Sir William Crookes. Crookes concluded that the cathode ray was made of particles which must have mass. Notice that the cathode and anode are positioned so that the rays will strike the top of the paddle wheel. Upon starting up the cathode ray tube, the wheel rotated from the cathode towards the anode. Attached to the rail was a paddle wheel capable of rotating along the rail. A cathode ray tube was constructed with a small metal rail between the two electrodes.The shadow caused by the object indicates that particles were being blocked on their way from the cathode to the anode. When an object was placed between the cathode and the opposite end of the tube, it cast a shadow on the glass.He discovered two things which supported the hypothesis that the cathode ray consisted of a stream of particles. Crookes also carried out many experiments using more reliable equipment to confirm earlier finding about the properties of cathode rays. His major contribution to construction of the tube was to develop ways to evacuate almost all the air from the tube. In 1878, Sir William Crookes, a British scientist, displayed the first cathode rays using a modification of the Geissler apparatus. Geissler explored a number of techniques to remove air from the tube and to prevent leaks, as well as ways to get good connections of the wires in the tubes. He used a mercury pump to create a vacuum in a tube. The first cathode ray tube prototype was developed by Heinrich Geissler, a German glassblower and physicist. 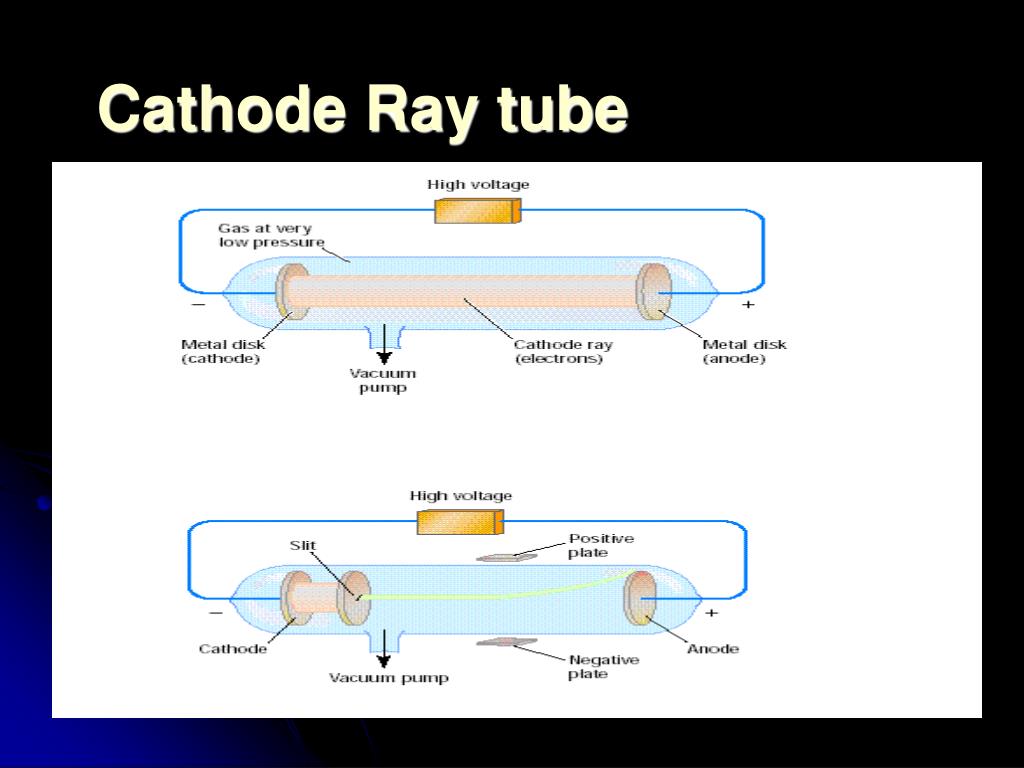
Similar CRT (cathode ray tube) devices were used in computer monitors, now also replaced by flat screen monitors. A beam of electrons was sprayed to a picture tube which was treated to react with the electrons to produce an image. The technology used in the older TV sets used cathode ray tubes. The new TV sets are flat screen technology that take up less space and give better picture quality, especially with the advent of high-definition broadcasting. 
The main reason is because they are older and based on outdated technology. The TV set seen below is becoming harder and harder to find these days. His work on electrolysis paved the way for subsequent experiments performed using cathode ray tubes. Faraday ultimately became the first and foremost Fullerian Professor of Chemistry at the Royal Institution, a lifetime position. His main discoveries include the principles underlying electromagnetic induction, diamagnetism and electrolysis.Īs a chemist, Faraday discovered benzene, investigated the clathrate hydrate of chlorine, invented an early form of the Bunsen burner and the system of oxidation numbers, and popularized terminology such as "anode", "cathode", "electrode" and "ion". Faraday (1791–1867) was an English scientist who contributed to the study of electromagnetism and electrochemistry. Davy is supposed to have even claimed Faraday as his greatest discovery. He also studied the forces involved in these separations, inventing the new field of electrochemistry.ĭavy's laboratory assistant, Michael Faraday, went on to enhance Davy's work and would become the more famous and influential scientist. The following year, he discovered calcium, strontium, barium, magnesium and boron, as well as the elemental nature of chlorine and iodine. He went on to electrolyse molten salts and discovered several new metals, including sodium and potassium in 1807. He was a pioneer in the field of electrolysis, which uses the voltaic pile to split common compounds and thus prepare many new elements. Sir Humphry Davy, 1st Baronet (1778–1829) was a Cornish chemist and inventor.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
